Nematodes - rice
Contributers to this page: IRRI, Seed Health Unit, Los Banos, Philippines(Patria Gonzales, Evangeline Gonzales, Carlos Huelma, Myra Almodiel, Joel Dumlao).
White tip
Scientific names
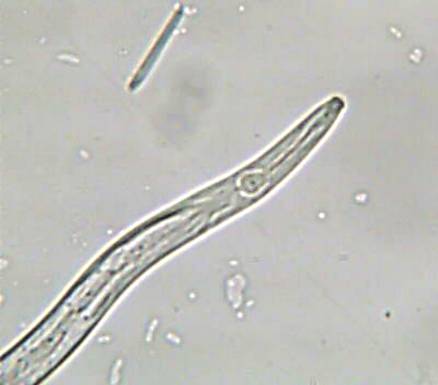
Aphelenchoides besseyi Christie
Significance
Yield of infected plants is lost or reduced but yield lost or reduction vary depending on cultivar, year, temperature, and cultural practices
According to reports, average yield losses in infested fields ranged from 10-30%;in fields where all plants (most susceptible cultivars) have been attacked, yield losses were up to 70%
Severity of the disease is increased by Ammonium sulfate, ammonium nitrate, calcium superphosphate, and potassium chloride.
Symptoms
Whitening of 3-5 cm of the leaf tip which eventually become necrotic and shred (typical symptom). The central part and the base of the infected leaves are darker green than normal. The upper leaves are the most affected, with the flag leaf often twisted, hindering the emergence of the panicle.
Other symptoms-reduced panicle length, reduced number of grains, sterile flowers, deformed grains, stunted plants, late ripening and maturation of grains, production of tillers in the upper nodes
Not all plant infected with A. besseyi show the white tip symptom although the yields are reduced
Host range
Setaria viridis (foxtail), Panicum sanguinale (crab grass), Cyperus iria, Cyperus polystachyus, Imperata cylindrical
|
|
Also known to occur in millet, strawberry, Vanda orchids, chrysanthemum, saintpaulia, Chinese cabbage, onion, soybean, sugarcane, sweet corn, sweet potato, yam, and more than 35 genera of higher plants and many species of fungi (Fortuner & Williams (1975)
Geographic distribution
White tip has been reported in most of the rice-growing countries of Africa, North, Central, and South America, Asia, Eastern Europe, and the Pacific.
Biology and transmission
A. besseyi parasitizes flooded and upland rice. This nematode survives from one season to the next in infected seeds; can survive in seeds in a dehydrated state up to 3 years; can also survive to a lesser extent on weeds, on ratoons, and debris from the preceeding rice crop.
Reviviscent nematodes that emerge from the seeds after planting are attracted by and feed on the primordial of emerging plants, later moving to the growing points of the stem and leaves feeding ectoparasitically. The nematode multiplies abundantly and is carried upward as the plant grows. The nematodes eventually migrate to the developing panicle, punctures the inflorescence, and penetrates into the florets via the apicular opening, where it feeds (on the ovaries, stamens, and embryos) and reproduces. After anthesis, reproduction declines and ceases in maturing grains, becoming dormant as the grains dehydrate.
The nematode is inactive if the atmospheric humidity is less than70% and migrates on aerial plant parts when they are covered with a film of water. The optimum temperature for development is 23-32°C but the nematode is active from 13-43°C. The nematode is killed at temperatures exceeding 43°C.
The nematode is effectively spread by irrigation water.
A. besseyi is amphimictic, but parthenogenetic reproduction may occur; life cycle takes 7-15 days depending on temperature and humidity; there are several generations during a growing season
Detection/indexing method in place at the CGIAR Center
- At IRRI – Modified Baermann Funnel Method (Sedimentation Test)
Treatment/control
- Hot-water treatment of infected rice grains (52-57°C for 15 minutes; for large volumes of seeds, presoaking in cold water for 3 hrs, then, the hot water treatment). The treated seeds are re dried back to recommended 14 % MC Germination of some rice varieties maybe adversely affected, thus, it is best to plant seeds immediately after treatment.
Procedure followed at the centers in case of positive test
General Protocol:
- Seedlots positive with Aphelenchoides besseyi are subject to deletion, replacement, or hot water treatment taking into account the import regulations of country of destination
Note: All imported seedlots are planted in IRRI’s post entry plant quarantine area only. Plants showing disease symptoms are immediately destroyed and disposed according to the recommendation of the Philippine Plant Quarantine Service
References and further reading
Ou SH. 1985. Rice Diseases 2nd ed. The Commonwealth Mycological Institute. UK
Mew TW, Misra JK. 1994. A Manual of Rice Seed Health Testing. IRRI.
Webster RK, Gunnel PS, editors. 1992. Compendium of Rice Diseases. The American Phytopathological Society. USA
Comments
- No comments found





Leave your comments
Post comment as a guest