Conservation of cassava genetic resources
Contributors to this page: CIAT, Colombia (Daniel Debouck); IITA, Nigeria (Dominique Dumet); Bioversity International/ILRI, Ethiopia (Alexandra Jorge); independent consultant (Clair Hershey); INIA, Peru (Llerme Rios).
Importance of cassava conservation
The potential for genetic improvement of any crop relies on the ability to successfully use the existing genetic resources, including the related wild species. Cassava is the fourth most important food calorie crop in the tropics, and is growing in importance both for food security (especially Africa) and in multiple commercial and industrial uses (mainly Latin America and Asia). The current cassava genetic resources are primarily landrace varieties of Manihot esculenta and wild Manihot species that evolved under natural selection, as well as farmer conditions (cultivated cassava) for centuries or even millennia. In the only comprehensive monograph on the genus, Rogers and Appan (1973) identified 98 species in the genus. Most of the genetic diversification occurred in the Americas where the genus originated, but secondary centers of diversity also exist in Asia and Africa.
It is estimated that nearly 2/3 of the genetic diversity exists only in situ, with only 1/3 being securely conserved ex situ. However, these estimates are based on expert opinion rather than on solid survey evidence and genetic analyses (Hershey, 2008).
- There are still many unique native landrace varieties to be collected and safely conserved.
On a global basis, cassava has relatively few improved varieties relative to many other important crops, since there are very few fully-fledged breeding programmes in the world, and most of these operate with very constrained resources. Most cassava germplasm in use is still based on landrace varieties. However, the adoption of new improved varieties has increased rapidly since the turn of the 21st century. In some cases, this adoption of new varieties is creating a risk for loss of native landraces.
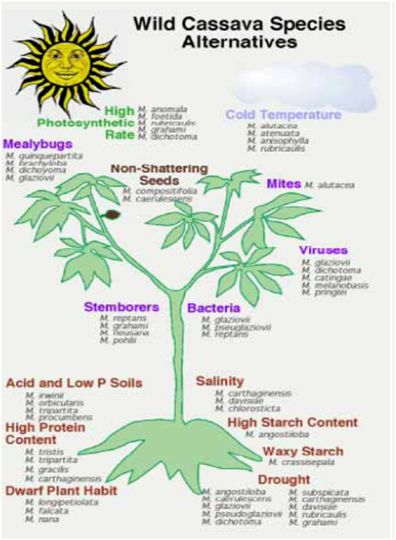
|
Figure 1 (Source: CIAT cassava programme) |
- Overall, farmers are adopting new varieties faster in Asia than elsewhere, especially in areas with high industrial demand. Landraces may be neglected and lost when adoption rates are high. Nonetheless, the areas where adoption rates are highest – China, Thailand and Vietnam – have very few landrace varieties and the development of new hybrids based on germplasm from the Americas is in fact introducing greater genetic diversity compared to what existed previously.
- In Africa, new varieties have been adopted due to increasing vulnerability of landraces to pests and diseases and higher productivity of new varieties. Landraces tend to disappear and many countries do not have adequate genebank conservation facilities.
- Expansion of agriculture and urbanization in South and Meso-America are shrinking the habitats of wild Manihot species.
- Wild species have played a limited but important role to date in cassava breeding programmes. However, the new challenges of cassava breeding, especially considering the new traits that will be needed for adaptation to climate change, and for new markets, are creating demand for new gene sources. This emphasizes and supports the need to collect and conserve wild types. To date there has been limited evaluation of the wild species for traits that may be relevant to the future of cassava breeding. Major efforts are needed to collect and conserve the Manihot species, especially those in areas where urbanization and agricultural expansion are threatening native habitats, such as in central Brazil.
- Figure 1 illustrates examples of the many possible traits that might be derived from wild Manihot species. However, it should be noted that most of these traits are hypothetical, as proper evaluations have not yet been done for the most part.
Conservation methods
Cassava
The combinations of genes currently found in the cassava landraces are a result of many centuries of farmers’ selection. Since cassava is a highly heterozygous crop, the only way to preserve these specific gene combinations is through vegetative propagation. In all cases, when seeds are produced from a particular landrace accession, there is broad segregation of the progeny due to the heterozygous nature of the parents. If accessions are to be recommended and used directly as new varieties by farmers, then it is critical that these specific gene combinations are preserved, for the desired traits to be expressed. However, if accessions are to be used by breeders as sources of specific genes, then the specific gene combinations are less important. In this case, seed conservation may be as good as, or better than, clonal conservation.
- The most widely used method of cassava conservation is through vegetative methods.
- Germplasm can be maintained vegetatively in fields or screenhouses (field banks) or in vitro (slow growth tissue culture or cryopreservation).
- When the objective is solely to conserve the genes (but not specific combinations of genes), true seed can be used instead, as long as flowering occurs. Seeds can also be conserved at low temperature and humidity or with cryo techniques.
- Genes can also be maintained for further use in the form of DNA (DNA banks) or cryopreserved pollen.
Genebanks need to have clear priorities as to which type of germplasm should be collected, efficiently conserved and securely duplicated.
- Small genebanks may be able to effectively and economically conserve all material, including accessions introduced from other countries and bred material.
- Larger genebanks should prioritize the conservation of local landraces, especially if resources do not allow adequate conservation of the entire collection that includes introduced material that is simultaneously conserved in other genebanks.
- If core collections have been defined, these may be given the highest priority, especially in terms of making material available for evaluation and breeding. Core collections are typically relevant only for large collections and there are only a few of these worldwide.
- Conservation of breeding lines and introductions should typically have lower priority than local material.
The conservation of local landraces in particular must be secured. Additional security can be provided by the following procedures:
- Duplicate in vitro collections in separate locations (preferred).
- Maintain a field bank and an in vitro bank.
- Use two separate field locations (least preferred).
Wild species
The wild Manihot species are conserved mainly in seed genebanks or as plants in the field. Since they are perennials, field genebanks may be maintained for long periods without regeneration if conditions are appropriate. Generally, the wild Manihot genebanks are planted or regenerated using seed. Some of the species have been successfully conserved and propagated in vitro; however each species appears to have unique optimum media and environmental conditions, and the research to define the suitable conditions has been done for only a relatively few species.
Major cassava collections
The largest ex situ cassava collections are held by CIAT-Colombia with about 6500 accessions, followed by EMBRAPA-Brazil (about 4000 accessions). Other important genebanks are those in CTCRI-India , INIA-Peru, NRCRI-Nigeria, IITA-Nigeria, IAN-Paraguay, SRCV-Benin, D.R. Congo and PGRC/CRI-Ghana.
The exact number of unique accessions can only be estimated, since there has not been an attempt to identify duplicate accessions across genebanks. There are probably more than 10 000 unique accessions conserved ex situ, in the more than 70 cassava genebanks worldwide (Hershey, 2008). Although there are no scientifically- or survey-based estimates, Hershey (2008) estimated more than 26 000 unique cassava landrace varieties, meaning that there are more uncollected landrace varieties than there are accessions already conserved ex situ. Clearly not every distinct genotype needs to be collected in order to capture the range of genetic diversity of the species. Hershey (2008) estimated that about 14 000 accessions, appropriately selected for genetic diversity, need to be collected and conserved to assure the long-term accessibility to the full range of genetic diversity of Manihot esculenta, and avoid genetic erosion of the species.
Many genebanks have part of their collection in tissue culture (in vitro). Among the major collections, only the CIAT genebank conserves the the collection exclusively in tissue culture or in cryo conditions. A few genebanks (mainly EMBRAPA, Brazil and CIAT, Colombia) have seed banks to conserve seeds of wild species or breeding material. A few are also initiating DNA banks.
CIAT and IITA are the two CGIAR centres sharing the international mandate for cassava research, breeding and improvement. They have been disseminating thousands of in vitro plantlets of improved varieties to dozens of countries through collaborating NARs or the private sector and development agencies. In addition, a very wide range of genetic diversity has been introduced from the CGIAR genebanks into national programmes through seed introductions. However, this is typically done by breeding programmes and does not necessarily become part of the cassava genebank.
The wild Manihot species have been given relatively little attention in terms of systematic collection and ex situ conservation. Firstly, many populations of these species have not been sampled and, secondly, many collected accessions have been lost due to difficulty in establishing and maintaining them.
References and further reading
Allem AC. 1992. Manihot germplasm collecting priorities. In: Roca WM, Thro AM, editors. Report of the First meeting of the International Network for Cassava Genetic Resources, 18-23 August 1992. Centro Internacional de Agricultura Tropical, Cali, Colombia. pp. 87-110.
Bonierbale M, Guevara C, Dixon AGO, Ng NQ, Asiedu R, Ng SYC. 1997. Cassava. In: Fuccillo D, Sears L, Stapleton P, editors. Biodiversity in Trust. Cambridge University Press. pp. 1-21.
Debouck DG. 1991. Genetic variation in crop species and their wild relatives: a viewpoint for their conservation. In: Becker B, editor. Genetic diversity and crop strategies for roots and tubers. Arbeitsgemeinschaft Tropische und Subtropische Agrarforschung e.V. and International Board for Plant Genetic Resources, Bonn, Germany, pp. 41-51.
Epperson JE, Pachico DH, Guevara CL. 1996. The cost of maintaining genetic resources of cassava, Manihot esculenta Crantz. Acta Horticulturae 429:409-413.
Epperson JE, Pachico DH, Guevara CL. 1997. A cost analysis of maintaining cassava plant genetic resources. Crop Science 37(5): 1641-1649.
Guevara C, Ospina JA, Mafla G, Verdier V. 1998. Zygotic embryo culture of Manihot esculenta Crantz: a practical approach for the safe international movement of cassava seed stocks. FAO/Bioversity Plant Genetic Resources Newsletter 115:33-38.
Gulick P, Hershey C, Esquinas AJ. 1983. Genetic resources of cassava and wild relatives. Series AGPG:IBPGR/82/111. IBPGR Secretariat, Rome. 60 pp.
Hershey C. 2008. A global conservation strategy for cassava (Manihot esculenta) and wild Manihot species. Summary of stakeholder deliberations and recommendations prepared for the Global Crop Diversity Trust. Available from: http://isa.ciat.cgiar.org/urg/urgweb_folder/files/unitfiles/A%20Global%20Conservation%20Strategy%20for%20Manihot%20%20August%202008-2.pdf. Date accessed: 7 Oct. 2010.
Hershey C, Iglesias C, Iwanaga M, Tohme J. 1994. Definition of a core collection for cassava. In International Network for Cassava Genetic Resources. Report of the First Meeting, CIAT, Cali, Colombia, 18-23 August 1992. International Crop Network Series No. 10, IPGRI, Rome, Italy. pp. 145-156.
Hidalgo R. 1995. Conservación Ex-Situ. In: "Memorias. Curso de Documentación de Recursos Fitogenéticos". Auspiciado por Universidad Nacional de Colombia, Bioversity y CIAT. Palmira. pp. 33-41.
Koo B, Pardey PG, Debouck D. 2004. CIAT genebank. In: Koo B, Pardey PG, Wright BD. Saving seeds: the economics of conserving crop genetic resources ex situ in the future harvest centres of the CGIAR. CABI Publishing, Wallingford, UK.
Leyton M. 1993. Crioconservación de pollen de yucca. Bachelor’s thesis. Univ. del Valle, Facultad de Ciencias, Dept. de Biología, Cali, Colombia.
Nassar NMA. 2006. Cassava genetic resources: extinct everywhere in Brazil. Genetic Resources and Crop Evolution 53:975-983.
Orrego JI, Hershey C. 1984. Almacenamieno del polen de yuca (Manihot esculenta Crantz) por medio de liofilizacion y varios regimenes de humedad y temperatura. [Storage of cassava (Manihot esculenta Crantz) pollen by means of lyophilization and various temperature and humidity regimes]. Acta Agronomica (Colombia) 34:21-25.
Patiño VM, Hershey CH. 1981. Collection priorities for cassava (Manihot esculenta) and wild Manihot species in Latin America. Series AGPG:IBPGR/81/95. IBPGR, Rome, Italy.
Perry M. 1994. Documentation and databases for germplasm collections. In International Network for Cassava Genetic Resources. Report of the First meeting, CIAT, Cali, Colombia, 18-23 August 1992. International Crop Network Series No. 10, IPGRI, Rome, Italy. pp. 157-162.
Rogers DJ, Appan SG. 1973. Manihot, Manihotoides (Euphorbiaceae), Flora Neotropica, Monograph no. 13. Hafner Press, New York. 272 pp.
Unnikrishnan M, Easwari Amma CS, Sreekumari MT, Sheela MN, Mohan C. 1987. Cassava germplasm conservation and improvement in India. In Proceedings of a cassava workshop in Asia. February 1987. Available from: http://webapp.ciat.cgiar.org/asia_cassava/pdf/proceedings_workshop_02/87.pdf. Date accessed: 26 August 2010.
Comments
- No comments found





Leave your comments
Post comment as a guest